
To summarize everything in this article, the following are some important points: When electrons are shared equally between atoms in a diatomic molecule or when polar bonds in a larger molecule cancel each other out, nonpolar molecules form (More details: Polar vs nonpolar molecules) Summary Polar molecules are formed when the electronegativity of the bonded atoms differs. Similarly, each of the three Br-F has a dipole with a non-zero value. It occurs in a liquid form with a strong odor at typical temperature and pressure conditions.Due to the existence of three fluorine atoms linked together, it is a strong fluorinating agent.It reacts vigorously with water and other organic compounds.The electronic repulsion occurs because the bromine atom contains lone pairs on it.Īccording to the VSEPR theory, the repulsion between the lone pair and bond pairs creates a downward force on the Br-F bonds, bending the form of the molecule. The bromine atom occupies the center position due to its lower electronegative nature. The fluorine atom has an electronegativity of 3.98, while the bromine atom has an electronegativity of 2.96. 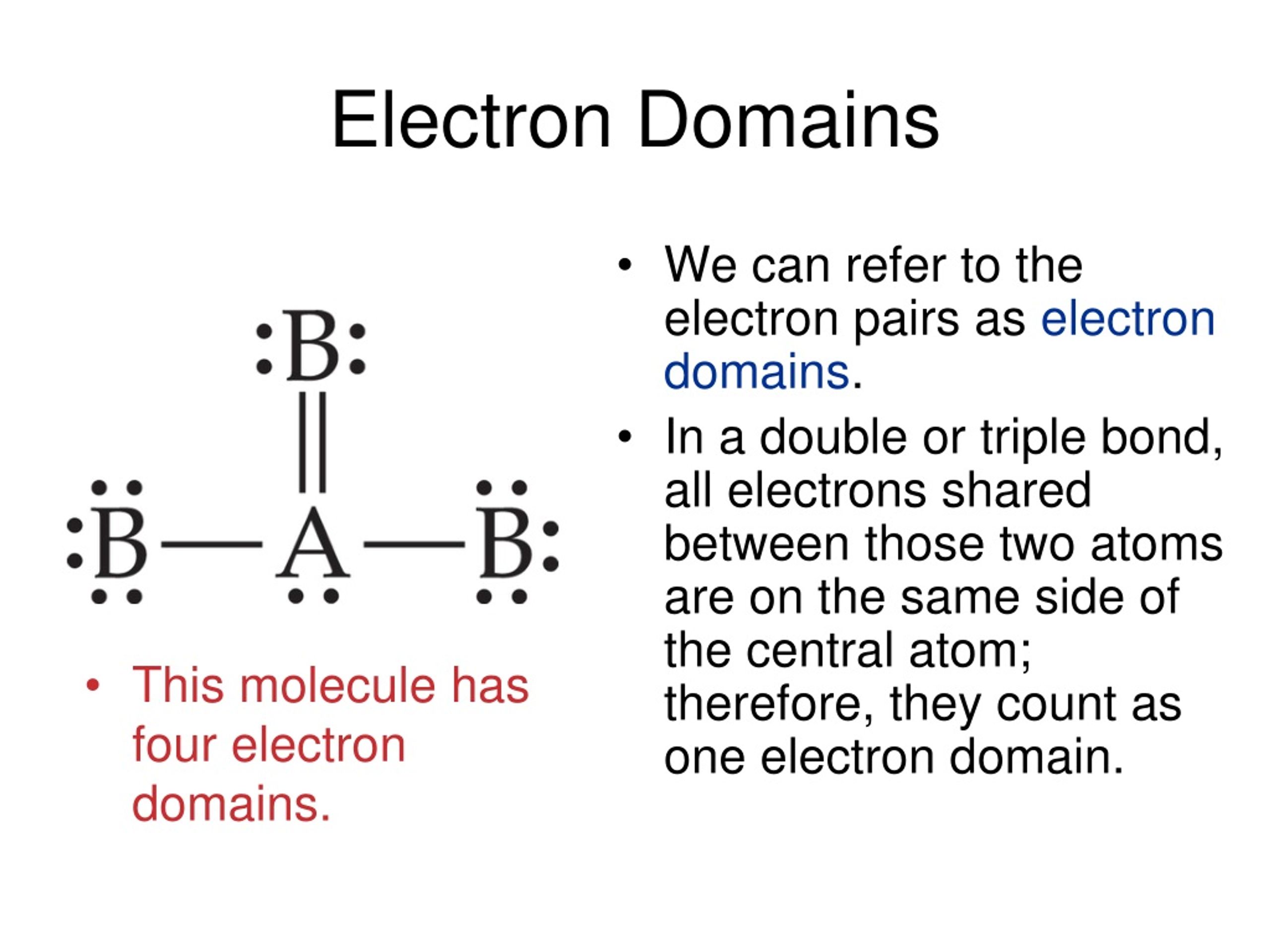
Three fluorine atoms surround one bromine atom in bromine trifluoride. The polarity across the Br-F bond is generated by the difference in electronegativity of bromine and fluorine atoms, with bromine as the positive pole and fluorine as the negative pole. The repulsion created by the electron pairs is higher than that of the Br-F bonds, resulting in this angle.īecause the bromine atom has two lone pairs, the electrical repulsion between lone pairs and bound pairs produces shape distortion, resulting in the bent shape. BrF3 has a T-shaped or Trigonal Bipyramidal molecular geometry, with a bond angle of 86.2 °, which is somewhat less than the typical 90°.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
